by EJ Ledet and Richard Bauman
Editor’s Note: This is the full scientific article that was abridged in our print issue.

The above photo depicts Lake Whatcom on a late summer evening at sunset in Sudden Valley. photo: EJ Ledet
What a beautiful lake and great place to live. We who live around the lake, along with the City of Bellingham, Whatcom County and our friends, neighbors, and nearby residents, enjoy the lake and draw drinking water from this lake. However, we are all responsible about the lake’s health and must do whatever is needed to preserve and improve its water quality.
Of particular concern is the lake’s declining amount of dissolved oxygen, which is crucial to its health. As the amount of dissolved oxygen (DO) declines, so does the lake’s water quality.
Solutions to that problem require understanding how oxygen enters the water and what causes its depletion.
This is a complex issue of multiple factors. Some physical conditions increase oxygen solubility which means dissolved oxygen reaches all depths of the lake – a good thing. But there are other contributors that change that solubility or even minimize or prevent it. There are also biological and chemical causes which decrease, deplete or consume dissolved oxygen and biological and chemical causes in the lake sediment which cause the lake’s health (water quality) to worsen.
We need to understand all these causes to be able to analyze cause and effect relationships and identify, develop and implement solutions to improve and maintain Lake Whatcom water quality.
We use a problem solving, Causal Analysis technique to define the problems, analyze their cause and effects and develop, fact-based cost-effective solutions.
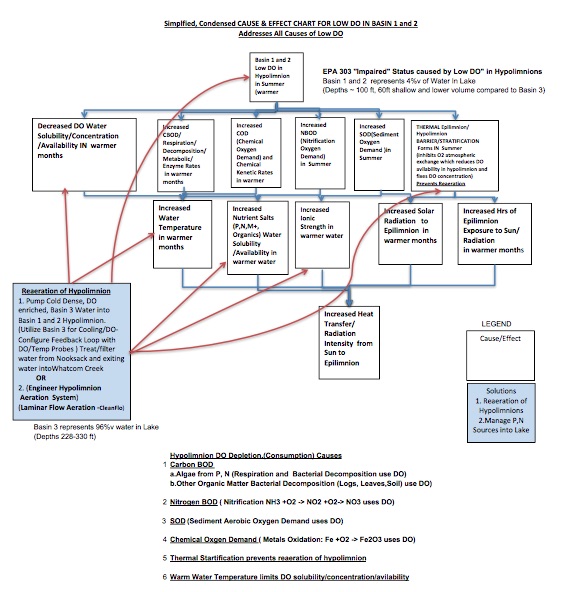
A Cause and Effect diagram (an evidence/fact based logic tree) begins with the problem definition/statement (Primary Effect) which needs to be very specific describing the what (Low DO), where/location (Basin 1and 2 hypolimnion), when/time (Warmer months – Summer), and Significance (EPA 303 Impaired Body of Water).
Once the problem is defined, the investigators begin asking “Why or Caused by” which leads to logic based development of a divergent (branched) series of causes and effects depicted as a chart/diagram. Investigators keep asking why or caused by until a sufficient number of causes and effects have been determined (minimum of 5 Whys).
Each Cause/Effect is supported with fact based, sensed evidence. Causes can be condition causes (occur over an extended period of time) or action causes (over a shorter period of time).
Figure 1 depicts a Simplified and Condensed Cause and Effect Chart/Diagram which shows the major causes for Low O2/DO in Basins 1 and 2. Basins 1 and 2 represent 4 percent of the total volume of water in Lake Whatcom.
Cause/Effects are depicted in each “white box.” Blue arrows point down and depict answers to “why or caused by.” Potential Solutions are depicted in “blue boxes.” Red arrows from the solutions box are drawn pointing to the cause or causes which they address
Problems occur when action and condition causes line up at the same time and in the same place/space. Effective solutions address the majority of causes and prevent, change, mitigate the primary effect/problem from recurring. The most effective solution(s) address(es) the action causes.
The Low DO Cause and Effect Diagram shows: 1. Physical causes of Warmer Water Temperature and Thermal Stratification; 2. Biological and Chemical causes of Carbon Biochemical Oxygen Demand -Algae and other organic matter (CBOD), Nitrification BOD (NBOD), Sediment Oxygen Demand (SOD), and Chemical Oxygen Demand (COD- i.e., metals oxidation).
How does O2/DO get into the Lake and how is DO transported from epilimnion to hypolimnion? What conditional causes effect DO solubility and DO re-aeration of the hypolimnion?
Past Western Washington University (WWU) and Washington Department of Ecology (DOE) water studies used data on phosphorous, algae/chlorophyll and low dissolved oxygen in Lake Whatcom to drive regulations to manage phosphorus that also increased property taxes and assessed fees in newly created storm water districts. A DOE statement: “Researchers have determined that excess phosphorous in the lake is the main cause of declining oxygen levels. This study quantifies how much phosphorous the lake can process naturally and still supply enough oxygen to meet state standards.”
The Environmental Protection Agency recommends using Causal Analysis Diagnosis Decision Information System, or CADDIS, to help scientists and engineers in the regions, states, and tribes conduct causal assessments in aquatic systems. The system basically focuses on where a problem occurs vs where it does not occur, or when a problem occurs vs when it does not occur, etc.
1. Physical Causes of Low DO in Basins 1 and 2
Warm lake water temperatures are caused by solar radiation intensity. Longer summer days increase the exposure, usually causing a decrease in oxygen solubility and concentrations of dissolved oxygen. Large and deep lakes like Lake Whatcom have layers of water, called strata, with different properties usually defined by water temperatures and density. This is how it works.
Thermal stratification is usually seasonal with clear delineations between layers (the scientific names are: upper – epilimnion, middle -metalimnion, bottom- hypolimnion) during the summer. During warmer weather months, heat transfer from the sun through conduction, convection, and radiation changes the temperature and water density in the middle stratum which, in turn, causes one or more barriers or thermoclines to form. These barriers of temperature and water density prevent a natural upwelling and exchange (scientifically, conduction and convection) between the strata and reduces aeration of and oxygen levels in the lower stratum.
Such problems tend to disappear in winter, when colder water temperatures foster mixing of the levels.
The major source of dissolved oxygen in Lake Whatcom in winter is the exchange between air and water (i.e., Aeration) and early spring mixing caused by wind, current, convection/conduction and turbulence.
An April 2017 article published in Whatcom Watch, “Lake Whatcom Update: Decline of Water Quality Accelerates,” by Dr. Matthews et al. (2016), lists several 2015 anomalies: “the abnormal results they measured in the lake last year were probably caused by two factors: 1) the warm weather from February through July 2015, which raised surface water temperatures throughout the lake to be higher than any measured in the past 30 years, and 2) the water in Basin 3 did not completely mix from top to bottom during the winter months for the first time ever recorded, probably due to weak winter storms and the shape of the basin. This caused oxygen deficits in the bottom waters from 2014 to remain and get worse throughout 2015.”
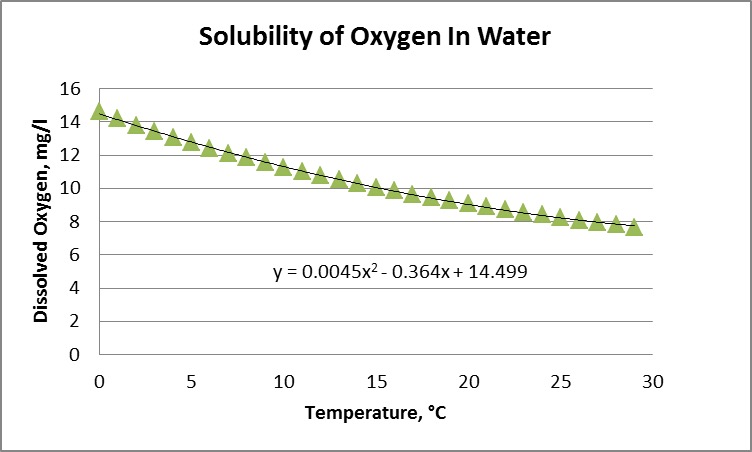
Warmer water temperature causes lower DO solubility/concentration/availability.
Chart 1 shows the relationship of oxygen solubility and water temperature. Colder water yields higher levels of dissolved oxygen.
DO water solubility is affected by temperature, pressure, and salinity. Lake Whatcom is a freshwater lake, so salinity isn’t a factor. Atmospheric pressure is of marginal impact due to the lake’s slight altitude of 310 feet above sea level. Which leaves water temperature as the key factor determining oxygen levels.
The article added: “The mild winter plus warmer spring and early summer weather, caused the lake to thermally stratify into separate layers sooner with warmer, less dense, oxygenated water in the upper layers above colder, denser water below. Biological processes in the isolated deeper water quickly consumed the dissolved oxygen, creating unoxygenated water in Basins 1 and 2 and oxygen deficits in Basin 3.”
And the longer such stratification in summer, the shorter the atmospheric exchange in winter and lower dissolved oxygen levels deep in the lake.
Longer duration period of Thermal stratification during warmer months can cause shorter atmospheric exchange time/period in winter. This, in turn, causes even lower initial DO concentrations in the hypolimnions.
Warmer water caused by longer warming periods cause lower DO water solubility. Extended Thermal Stratification times cause shorter DO atmospheric exchange (i.e., reaeration) in cooler months, in turn, causing even lower initial DO concentrations in the hypolimnions.
Dissolved oxygen in the deep strata is more quickly consumed by both biological and chemical processes. As dissolved oxygen levels decline, anaerobic bacteria/microbes increasingly decompose in oxygen-free sediment. Nitrates and nitrites are converted to ammonia. Sulfate salt, like iron sulfate is converted to iron sulfide and hydrogen sulfide. Insoluble ferric phosphate is converted to soluble ferrous phosphate. All these by products from sediment anaerobic decomposition can be released into the above water column, decreasing the overall health and water quality of the lake.
Biological and Chemical Causes of Low DO in Basins 1 and 2
Algae are typically named the villains in depleting oxygen in the lake. They feed on the phosphorus and nitrates in runoff that feeds the lake.
Algal respiration consumes dissolved oxygen at night, and bacteria and microbes consume when algae die and decompose.
But they’re not the only cause for low oxygen levels in the lake.
Organic matter carried by runoff and other sources also contribute. These include sawmill debris, sunken logs, coal mine debris, leaf litter, decomposing wooden train trestles and remnants of sunken boats deposited in lake bottom sediment. Bacteria and microbes aid in the decomposition of these and absorb oxygen through their digestion processes.
Solutions Development.
Effective solutions address the majority of causes and prevent, change, mitigate the primary effect/problem from recurring.
From – Current Lake: Decreased DO Supply + Increased DO Demand = Anaerobic (- DO) = BAD
To – Improve Lake: Increased DO Supply + Decreased DO Demand = Aerobic (+DO) = GOOD

Pumping system proposal for increasing dissolved oxygen in Basins 1 and 2. Courtesy: Mitchell et. al. 2010
Figure 2. Pumping System Proposal for increasing DO in Basin 1 and 2
Lake Whatcom is divided in three Basins, numbered from west to east. Basin 3 is the deepest, containing 96 percent of the lake’s volume and acts as a heat sink. So pumping water from its lowest stratum into the other shallower basins would generate the cold-water effects of the winter months. Basin 1 and 2 dissolved oxygen concentrations can be improved immediately by engineering a pumping feedback loop system with oxygen/temperature probes inserted at various depths in the lake. Pumping the denser, oxygen-rich water from the deeper Basin 3 into the bottom of Basins 1and 2 will displace the warmer, less dense and depleted water would be discharged via Whatcom creek.
A cooling and aeration system could be engineered to deal with snow melt, water entering from the Nooksack River diversion. The costs associated with implementing and managing such a pumping system will have to be determined but this solution could improve increase DO and perhaps remove the EPA 303 impaired body of water classification.
Such a system would decrease the water temperature in the low stratums of Basins 1 and 2, thereby immediately increasing the dissolved oxygen content, prevent thermal stratification and decrease algal and biological growth, among other changes observed and measured during winter months.
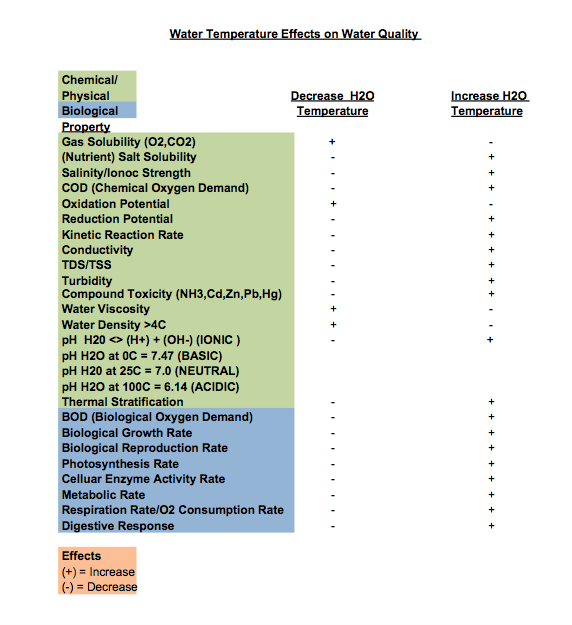
Table 1 was created from data listed in Fondriest Environmental, Inc.: Water Temperature.” Fundamentals of Environmental Measurements. 7 Feb. 2014. Web.
Solution 2. Install “Clean Flo” Hypolimnion Laminar Flow Aeration System
Laminar Flow Inversion and Oxygenation
The most important part of CLEAN-FLO’s unique water improvement process is called “Continuous Laminar Flow Inversion and Oxygenation.” CLEAN-FLO systems are designed to completely mix the surrounding waters and evenly distribute dissolved oxygen throughout the sediments for efficient microbial utilization. Continuous laminar flow inversion oxygenates the water and removes toxic gases. CLEAN-FLO process oxygenates an entire body of water from top to bottom.

Clean Flo diffuser which creates thousands of air bubbles at the bottom of the lake to re-oxygenate and de-stratify the lake. Courtesy: Clean Flo International
Figure 3. Clean Flo Diffuser creates tiny air bubbles. Set of diffusers oxygenate an entire body of water
Laminar flow created by CLEAN-FLO systems is non-turbulent and will not increase suspended solids or increase turbidity. In fact, the opposite is true, suspended solids and turbidity will be reduced. CLEAN-FLO diffusers are placed on the bottom and are not suspended above the sediments. As the bubbles release from a diffuser, oxygen is transferred to the water from the bubble, and they also move water gently to the surface and across the surface where additional oxygen is absorbed by the water. . This oxygenation helps purge the water of carbon dioxide (CO2), which produces an environment that promotes better water quality. Other gasses such as hydrogen sulfide (H2S) and ammonia (NH3) are also purged from the sediments.
 Laminar flow inversion and oxygenation carries oxygenated, toxic gas-free surface water down to the bottom where it initially binds phosphorus and nitrogen to the sediments and then reduces sediment P, and N via biochemical pathways of P,N metabolism in re-establishing the food chain, thus reducing P which the TMDL addresses. Oxygenation enables beneficial microorganisms to feed on bottom non-living organic sediment. It enables aquatic insects to feed on the microorganisms, and fish to inhabit the bottom waters and feed on the insects, providing a valuable natural food source to improve fish growth and health.
Laminar flow inversion and oxygenation carries oxygenated, toxic gas-free surface water down to the bottom where it initially binds phosphorus and nitrogen to the sediments and then reduces sediment P, and N via biochemical pathways of P,N metabolism in re-establishing the food chain, thus reducing P which the TMDL addresses. Oxygenation enables beneficial microorganisms to feed on bottom non-living organic sediment. It enables aquatic insects to feed on the microorganisms, and fish to inhabit the bottom waters and feed on the insects, providing a valuable natural food source to improve fish growth and health.

This ring is caused by Clean Flo, a system to solve the dissolved oxygen problem by completely mixing the surrounding waters and evenly distribute dissolved oxygen throughout the sediments. Courtesy: Clean Flo International
We asked Clean Flo for cost estimates of the Clean Flo System and proposed evaluating this proposed solution on a Pilot basis. Attached are the estimates. We would also propose installing/testing the pilot in Basin 2 first and then decide on forward strategy basis typical water quality results which could be performed by WWU laboratory.
Basin 2 Pilot ($850,000-$1,000,000 first year and $200,000 each year to operate/maintain after installation)
1. Cost (materials, labor, installation, travel, etc.) $650,000 not including site work, electrical and building to house compressor
2. Annual maintenance- electricity, analytical testing costs, augmentation chemicals, nutrient sponge, etc. $150 – 200,000
3. Installation timing (time of year; duration) – any time weather permitting – 2-3 weeks
4. How much time before we began see results/evidence that Clean Flo works in Basin 2? 30-60 days and will continue to improve for up to one year.
Basin 1 $820,000-$1,000,000 first year and $200,000 to operate and maintain each year after installation)
1. Cost (materials, labor, installation, travel, etc.) $620,000 not including site work, electrical and building to house compressor. Other cost estimates were the same as those in Basin 2..
Changing the predominant conditions in the water column (particularly in benthic zones) from anaerobic to aerobic is a primary change of system conditions that enables the biochemical pathways for nutrient metabolism to be directed into sustainably more beneficial outcomes. These include:
• Reduction of toxic cyanobacteria
• Reduction of the risk of cyanobacteria producing microcystin and other toxins when they are subject to algaecide treatment in the reservoir or chlorination in purification ic detritus etc.)
• Reduction in chemical costs associated with water treatment – less algae, lower disinfectant usage, lower THM formation (disinfectant by product carcinogens)
• Reduction in hazardous conditions (HABs, E. Coli etc.) that restrict recreational use of the water body.
Strengthening the food chain results in larger, healthier fish populations which attract predators such as eagles, pelicans etc.
Solution 3. TMDL P – Estimated price tag of $100 million dollars and 50-year period to bring dissolved oxygen concentration in Basins 1 and 2 to 2003 levels. “Spend $50 million in first 5 years, $10 million dollars per year to improve water quality” in Lake. TMDL P Modeling Assumes that main cause for DO depletion in Lake Whatcom is P which, in turn, causes Algae DO consumption via respiration and DO depletion via death/decay by aerobic bacteria/microbes (Carbon BOD). Although P contributes to organic and inorganic matter which is decomposed by biological/biochemical and chemical processes deposited on and in sediment, P represents a fraction of total (organic plus inorganic) matter which is composed of C, H, O, N.S, and P and other minor elements.
Also managing P from entering Lake Whatcom via TMDL P, by constructing natural filtration systems, will not address the P and other nutrients (N) which already exist in lake sediments.
We have raised concerns that the TMDL P solution will not address all causes of Low DO in Basins 1 and 2 and will not improve hypolimnion DO by itself.
Causal Analysis shows DO depletion/consumption in Basin 1 and 2 hypolimnion is caused by algae plus other organic matter aerobic CBOD causes, NBOD, SOD, COD, as well as water temperature and thermal stratification causes.
Holistically, all causes for DO depletion in Basins 1 and 2 can only be addressed by re-oxygenation of the hypolimnions, where Low DO occurs and where the 303d impairment was assigned/legislated.
DO can be improved by oxygenation of the entire water column, all layers, of Basin 1 and 2.
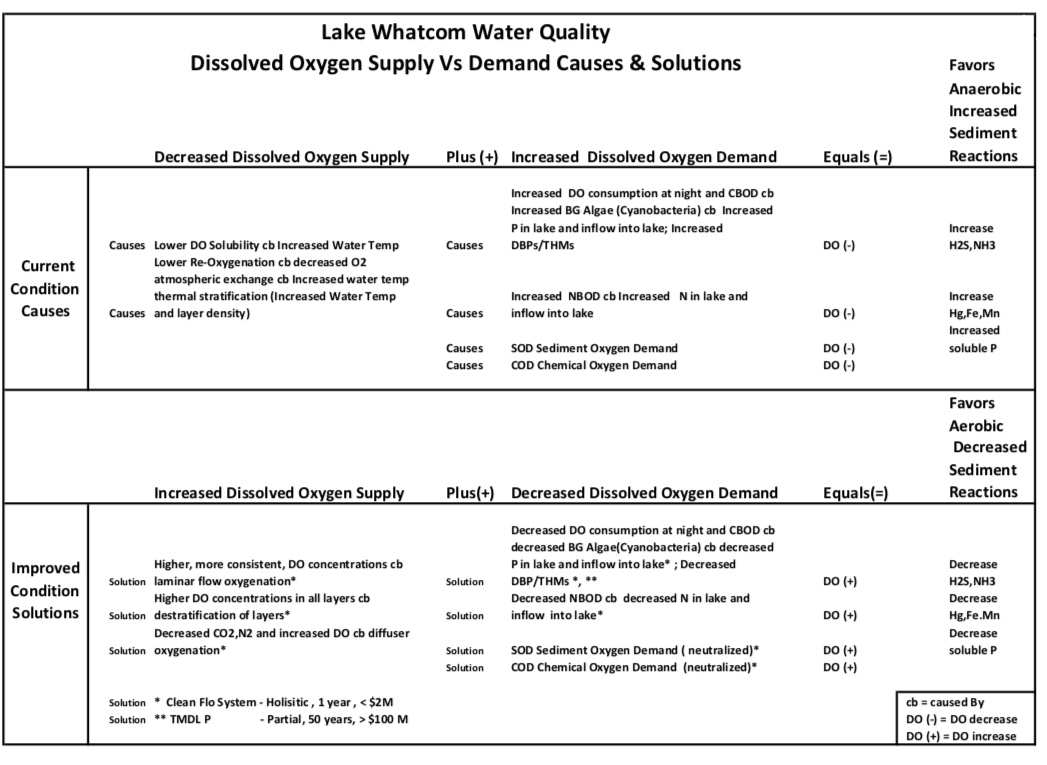
Table below depicts Lake Whatcom Dissolved Oxygen Supply Vs. Demand Causes and Solutions.
Table definitions:
Supply Oxygen Processes – DO water solubility and Atmospheric O2/DO – water exchange between all layers
Demand Oxygen Processes (Biological, Biochemical, Chemical processes which consume oxygen (aerobic) to decompose organic and inorganic matter in Lake
- CBOD – carbon biochemical oxygen demand (uses DO to breakdown organic (C) matter)
- NBOD – nitrification oxygen demand (uses DO to convert ammonia to nitrite and nitrate salts
- SOD – sediment oxygen demand (uses DO to breakdown other material in sediment
- COD – chemical oxidation demand (uses DO to convert metal to oxides like iron to form rust
Sediment Anaerobic Biochemical Processes – do not require DO but usually acquire DO/O2 through decomposition of different materials (e.g., Nitrate salts (NO3-) and sulfate salts (SO4-) release O2 to form NH3 and H2S; carbonate salts (CO2-) lose O2 to form methane (CH4).
Proposed Solution Scenario(s) to Implement the Most Cost-Effective Solutions to address Low DO Causes
Implement Clean Flo Pilot in Basin 2 for $850,000 in 2018. Evaluate Success and determine forward strategies for implementing Clean Flo in Basins 1 and eventually in Basin 3.
Table compares the Solutions for DO vs Water Quality Property and also includes estimated costs for each solution for Basin 1 and 2.
Low DO Solutions
We disagree with the statement/ interpretation that P is the primary cause of Low O2 in Basin 1 and 2. Although managing/minimizing P in Lake Whatcom Watershed via (TMDL P) will help reduce/eliminate Algae and minimize consumption of some of the Low DO (DO depletion) in Basin 1 and 2 hypolimnion, it will not address all causes of Low DO nor address the P and other nutrients already present in the lake.
We believe re-oxygenation of the hypolimnion via implementation of the most cost- effective reaeration solution coupled with a more cost effective TMDL stream and storm water management abatement plan is critical to restoring and improving DO concentrations in Basin 1 and 2.
The Clean Flo, Laminar flow aeration system, solution improves DO, captures and consumes P, N inflow into and already in the lake, effectively manages Algae and Chlorophyll, reducing water treatment costs, rebuilds a healthy food chain sustaining and promoting fish growth and reproductive rates, and prevents anaerobic biological and chemical reactions from occurring in the lake sediments (i.e., formation of hydrogen sulfide, ammonia, methyl mercury, iron, magnesium, and lead) . It is the most cost- effective solution for Lake Whatcom and will improve and enhance Lake and Drinking Water Quality and remove the EPA 303d impaired water body classification. Clean Flo will manage “how much phosphorous the lake can process naturally and still supply enough oxygen to meet state standards.”
Clean Flo manages phosphorous and nitrogen inflow into the lake as well as phosphorous and nitrogen already present in the lake and eliminates the need and costs associated with building/constructing natural soil drainage filters/catch basins.
I wrote numerous emails and spoke to several city officials (CoB, WC, City Planners, WWU researchers, DOE regulators) and sent them a rough draft of this article.
I heard back from Dr. Matthews via email. She thought Clean Flo was more appropriate for Eutrophic Lakes and was working on writing her annual report for the city and could not meet with me to discuss my article because she was too busy at the moment. I did not hear back from Steve Hood with DOE.
I did speak (3 mins) to WC council members on December 5,, 2017, Public Meeting to discuss Newly Formed Storm Water District Tax Assessment. I told members that I had concerns that the TMDL P would not solve the Low DO problem and did not address P already in the Lake. I presented the Low DO Solution comparison table and told them that re-aeration (re-oxygenation) using the Clean Flo solution was the most cost-effective and would solve and improve DO and other water quality issues.
One councilmen commented that aeration had been previously proposed and rejected but that it should be revisited. The main issue with other aeration systems is disturbing sediment, high turbulence, and mixing sediment in water column. Clean Flo does not disturb/mix sediment. In fact, it does the opposite.
I continue to send Clean Flo updates to CoB, WC, WWU, DOE. I am placing articles in SV Views, Whatcom Watch Online, SMLLibety Road Website, and have remitted my article for publication in Science Magazine and in WALPA.
I want to inform/educate taxpayers to implement the most cost- effective solutions to improve Lake Whatcom Source and Drinking Water Quality.
I do this freely as a concerned citizen, researcher, and taxpayer and because I want to add value to this beautiful area of the country.
References/End Notes:
1. https://www.epa.gov/caddis; https://www.epa.gov/sites/production/files/2015-12/temp-cd_sim_1500.jpg ;
https://www.epa.gov/caddis-vol1/caddis-volume-1-stressor-identification-step-2-list-candidate-causes
2. Whatcom Watch Online “Lake Whatcom Update: Decline of Water Quality Accelerates”, Dr. Matthews et al. (2016) http://whatcomwatch.org/index.php/article/lake-whatcom-update-decline-of-water-quality-accelerates/
3. Overview of Root Cause Analysis Techniques R. Bowen 5/18/2011
5. Minnesota Pollution Control Agency; Dissolved Oxygen TMDL-Protocols and Submittal Requirements, December 2008
6. Fondriest Environmental, Inc.: Water Temperature.” Fundamentals of Environmental Measurements. 7 Feb. 2014.
7. Identification of Fecal Escherichia coliform in Humans and Animals by Ribotyping, C.A. Carson, B. Shear, et al, Applied and Environmental Microbiology, December 2017,Vol 83, Issue 23
8. https://www.dropbox.com/s/2nr1rxu8u8j2ua2/Letter%2012-1-17.pdf?dl=0Clean Flo
9.https://www.dropbox.com/s/92gj04o237y9t0a/Lake%20Whatcom%20TMDL%202002%20Study%20.pdf?dl=0
_________________________________
E.J. (Enoch) Ledet has over 40 years experience as a chemist, biochemist, and causal analysis investigator/facilitator in the petrochemistry industry. He specialized in ensuring product integrity in both laboratory and field operations management and in the design and use of laboratory quality assurance systems.
Richard Bauman is a chemical engineer with 35 years experience working for Exxon- Mobil. He was the laboratory director responsible for research and development of gas conversion and shale retorting processes.